Dr David Murray
Principal Investigator/Senior Lecturer
Molecular Cell and Developmental Biology, School of Life Sciences

Contact
Biography
I am a protein biochemist focused on mechanistic cell biology of membrane-protein interfaces, inspired by the world of multicellular life. I have worked on molecular mechanistic questions since my PhD at University of Virginia, USA, where I focused on regulation of the membrane fusion machinery in model membranes. Following this period, I learned large protein complex biochemistry in the lab of Jim Hurley (at the NIH, Bethesda, USA) and subsequently joined Marino Zerial’s lab (Max Planck Institute for Molecular Cell Biology and Genetics, Dresden, Germany). I determined a molecular mechanism for extended coiled-coil membrane tethers, combining state-of-the-art biophysical and experimental cell biology approaches. In 2018, I established my programme in Dundee, supported by an independent Wellcome Trust fellowship. Since then, we have paved the way for broad impact in membrane biology. Our vision is to enable the engineering of cell form, through specific targeting of trafficking pathways.
Research
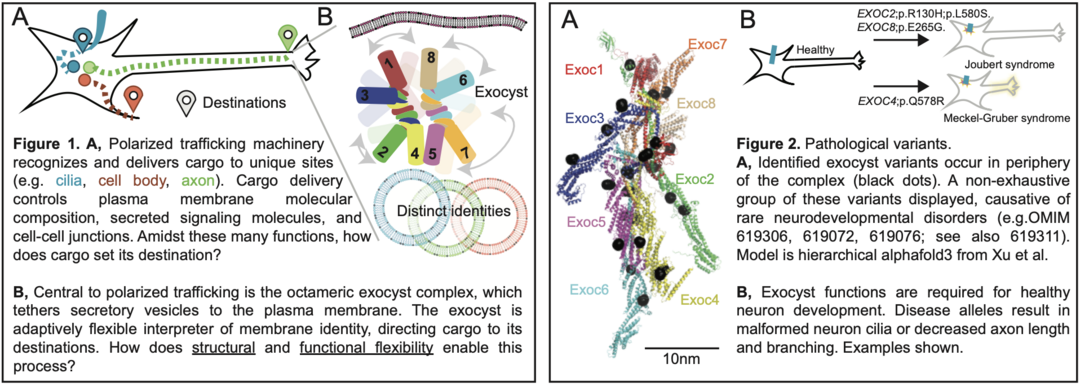
The cell biology of membrane trafficking and its links to cell organization, communication, and disease.
In multicellular organisms, the composition and organization of the plasma membrane defines essential cellular attributes. Membrane composition and architecture is organized by polarized trafficking, which delivers receptors, secretes signaling molecules, and organizes cell-cell connections. Perturbation of these functions results in dysregulation of the plasma membrane, and is associated with disease.
A striking example of the importance of polarized trafficking is neuron organization. Indeed, pathogenic variants in polarized trafficking components result in neurodevelopmental diseases. However, there is a critical knowledge gap between our understanding of the “parts list” of polarized trafficking, and its impact on plasma membrane organization.
How can a single, conserved machinery adaptively deliver cargo across unique cell types and morphologies? We hypothesize that a fundamental understanding of polarized trafficking in specialized cells will enable its targeted modulation. We study cell systems which undergo changes, just like in development or disease, to test this hypothesis and others. Our lab is interested in protein biochemistry, biophysical methods, cell biology, and physiological cell systems. We are interdisciplinary and ambitious in focus. Ultimately, we aim to reveal molecular models for the many specialized cellular functions of polarized trafficking, with the long-term aim to revert cell state dysfunction in disease.
Selected Publications
- Maib, H., and D.H. Murray. A mechanism for exocyst-mediated tethering via Arf6 and PIP5K1C driven phosphoinositide conversion. Current Biology, 2022 32(13):2821. https://doi.org/10.1016/j.cub.2022.04.089
- Vines, J., Maib, H., Murray, D.H., and J. King. A PI(3,5)P2 reporter reveals PIKfyve activity and dynamics on macropinosomes and phagosomes. JCB, 2023 222 (9): e202209077. https://doi.org/10.1083/jcb.202209077
- Maib. H., Adarska, P., Hunton, R., Vines, J., Strutt, D., Bottanelli, F., and D.H. Murray. Recombinant biosensors for multiplex and super-resolution imaging of phosphoinositides. JCB, 2024 223 (6): e202310095. https://doi.org/10.1083/jcb.202310095
- De la Concepcion, J.C., Duverge, H., Kim, Y., Julian, J., Xu, H.D., Ikene, S.A., Bianchi, A., Grujic, N., Papareddy, R.K., Grishkovskaya, I., Haselbach, D., Murray, D.H., Clavel, M., Irwin, N.A.T., and Y Dagdas. Electrostatic changes enabled the diversification of an exocyst subunit via protein complex escape. bioRxiv 2024.08.26.609756; https://doi.org/10.1101/2024.08.26.609756 (in revision)
- Xu, H.D., Badonyi, M., Sopipong, W., MacGowan, S.A., Marsh, J.A. and D.H. Murray. Structural extension of the human exocyst is enabled by a minimal interface. bioRxiv 2024.10.07.617058; https://doi.org/10.1101/2024.10.07.617058 (in revision)
- Loyer, N., Hogg, E.K.J., Shaw, H., Murray, D.H., Findlay, G.M. and J. Januschke. A CDK1 phosphorylation site on Drosophila PAR-3 regulates neuroblast polarisation and sensory organ formation. eLife, 2025. https://doi.org/10.7554/eLife.97902
PhD Projects
Principal supervisor
- Type
- PhD opportunity
Cryo electron microscopy of the exocyst complex on membranes
- Type
- PhD opportunity
Regulation of polarized trafficking amidst change
Awards
| Award | Year |
|---|---|
| Personal Fellowships / Royal Society and Wellcome Trust Sir Henry Dale Fellowship | 2018 |
Stories
- Type
- News
Wellcome Trust Early-Career Award for Hannes Maib
Dr Hannes Maib has been awarded a 5-year Wellcome Trust Early-Career Award that will allow him to establish his own independent laboratory at the University of Sheffield.
- Type
- News
£2 million boost for Dundee researchers
Two University of Dundee scientists have been awarded prestigious fellowships.
