
Contact
Research
Temperature shapes plant growth
Plants display a remarkable degree of developmental plasticity: they constantly adjust their growth and morphology in response to their surroundings to maximise fitness and reproductive success. Temperature is arguably one of the most influential factors impacting plant fitness as it affects the rate of every physical and biochemical reaction; it is therefore of vital importance for the plant to be able to sense its temperature environment. With the progression of climate change, plants in temperate regions such as the UK are challenged by more frequent heat waves as well as an overall rise in temperature, and these effects already impact crop production: in wheat and barley, each 1 °C increase above optimal growth temperature is estimated to reduce crop yield by 5-6%. Gaining a deeper understanding of how plants perceive temperature cues and translate them into appropriate developmental and physiological responses is thus a central question in both fundamental plant science and agriculture.
In my lab we mainly use the model plant Arabidopsis thaliana to understand the fundamental processes of plant temperature signalling (Figure 1A). In Arabidopsis, prolonged exposure to warm temperatures triggers a set of developmental responses that we collectively refer to as thermomorphogenesis: plants show increased elongation growth of the hypocotyl (embryonic stem) and primary root, an upward bending of the leaves, reduced formation of stomata (microscopic pores for gas exchange and evaporation) as well as accelerated flowering and seed set. In addition to Arabidopsis, we are also also looking to translate some of our research into barley (Figure 1B), one of the UK’s staple crops, whose development and yield are highly sensitive to ambient temperature.
Sensing temperature at the molecular level
Research in the past two decades revealed that the protein family of PHYTOCHROME INTERACTING FACTORS (PIFs) acts at the heart of temperature signalling in Arabidopsis. These transcription factors control expression of a large set of genes in a temperature-dependent manner and thereby affect many aspects of thermomorphogenesis. But expression of PIFs is itself highly temperature-sensitive (Figure 2): transcription of the PIF4 gene increases when temperatures rise, which, in addition with increased protein stability, leads to strong accumulation of PIF4 protein in the heat. In case of PIF7, we find that warm temperatures do not affect its transcription, but enhance its translation rate, i.e. how much protein is made from its transcript. This process is in part mediated by a thermosensitive RNA structure (RNA thermoswitch), which changes its conformation upon a temperature shift (Chung*, Balcerowicz* et al., 2020). Similar structures exist in other transcripts, but whether they fulfil a similar function is yet unknown.
Using a combination of genetics, molecular biology, biochemistry and omics approaches, we tackle the following questions:
- Which genes, alongside PIF4 and PIF7, control molecular responses to warm temperature?
- How do warm temperatures selectively affect the transcription and translation of temperature signalling genes?
- How can we harness the identified signalling mechanisms to render plants more resilient towards a changing climate?
Integrating light and temperature information in space and time
Temperature cues are not perceived in isolation but are integrated with a variety of other environmental and endogenous signals. In Arabidopsis, light and temperature sensing is particularly intertwined, using a largely overlapping set of genes including PIFs (Balcerowicz, 2020). Light and temperature jointly regulate downstream signalling programmes, and plant hormones such as auxin, gibberellin and brassinosteroids feature prominently among them. These downstream programmes are not static, but change over time and also differ across tissues and organs. The precise nature of these differences, and how they are established, however, remains largely unexplored.
Alongside whole-plant genetic and transcriptomic approaches, we use local genetic perturbations and bioimaging down to the cellular level to address the following questions:
- How do light and temperature responses differ across tissues and organs, both at the phenotypic and molecular level?
- What factors provide spatial and temporal specificity to light and temperature responses?

Figure 1: Arabidopsis (A) and barley (B) seedlings show developmental responses to warm temperatures.
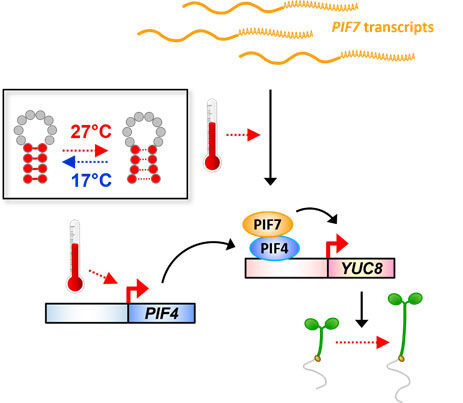
Figure 2: Temperature controls PIF gene expression. Warm temperature increases transcription of the PIF4 gene and enhances translation of the PIF7 transcript. The latter response is in part mediated through a structural change in the transcript’s 5’-UTR. Consequently, high levels of PIF4 and PIF7 protein activate thermomorphogesis genes such as the auxin biosynthesis gene YUC8.
Selected Publications
Research Articles
Balcerowicz, M.*, Mahjoub, M.*. Nguyen, D., Lan, H.. Stoeckle, D., Conde, S., Jaeger, K., Wigge, P., Ezer, D. (2021) An early-morning gene network controlled by phytochromes and cryptochromes regulates photomorphogenesis pathways in Arabidopsis. Mol. Plant 14: 983-996 (* shared first authors)
Chung, B.Y.W.*, Balcerowicz, M.*, Di Antonio, M., Jaeger, K.E., Geng, F., Franaszek, K., Marriot, P., Brierley, I., Firth, A.E., Wigge, P. (2020) An RNA thermoswitch controls daytime growth in Arabidopsis. Nat. Plants 6: 522-532 (* shared first authors)
Balcerowicz, M., Kerner, K., Schenkel, C., Hoecker, U. (2017) SPA Proteins Affect the Subcellular Localization of COP1 in the COP1/SPA Ubiquitin Ligase Complex during Photomorphogenesis. Plant Physiol. 174: 1314-1321.
Balcerowicz, M., Ranjan, A., Rupprecht, L., Fiene, G., Hoecker, U. (2014) Auxin represses stomatal development in dark-grown seedlings via Aux/IAA proteins. Development 141: 3165-3176.
Balcerowicz, M.*, Fittinghoff, K.*, Wirthmueller, L., Maier, A., Fackendahl, P., Fiene, G., Koncz, C., Hoecker, U. (2011) Light exposure of Arabidopsis seedlings causes rapid de-stabilization as well as selective post-translational inactivation of the repressor of photomorphogenesis SPA2. Plant J. 65: 712-723. (* shared first authors)
Review Articles
Balcerowicz, M., Shetty, K. N., Jones, A. M. (2021) Fluorescent biosensors illuminating plant hormone research. Plant Physiol. 187: 590-602.
Balcerowicz, M. (2020) PHYTOCHROME‐INTERACTING FACTORS at the interface of light and temperature signalling. Physiol. Plant. 169: 347–356.
Balcerowicz, M.# and Hoecker, U. (2014) Auxin – a novel regulator of stomatal development. Trends Plant Sci. 19: 747-749. (# corresponding author)
Awards
- Royal Society University Research Fellowship - 2022
- EMBO Long-Term Fellowship - 2016
Media availability
I am available for media commentary on my research.
Dr Balcerowicz is a plant molecular biologist with a specific interest in how plants perceive and react to their environment, particularly to light and temperature signals. His areas of expertise include plant development, plant genetics, light signalling, temperature sensing and plant responses to climate change.
Contact Corporate Communications for media enquiries.
Areas of expertise
- Climate change
PhD Projects
Principal supervisor
Awards
| Award | Year |
|---|---|
| Personal Fellowships / Royal Society University Research Fellowship | 2021 |
Stories
- Type
- News
Funding award to study how plants react to temperature
Dr Martin Balcerowicz has been awarded ~£540k to study the molecular mechanisms by which plants react to their temperature environment.
- Type
- News
Martin Balcerowicz joins Plant Sciences
Martin Balcerowicz joined the School in January to establish his own laboratory in the Division of Plant Sciences.