
Contact
Biography
Daniel Neill is Principal Investigator within the Division of Molecular Microbiology. He obtained a PhD in infection immunology from the MRC Laboratory of Molecular Biology, Cambridge, in 2010. He worked as a post-doctoral researcher, first at the University of Leicester, and later, from 2012, at the University of Liverpool. Whilst at Liverpool, he was awarded a Wellcome and Royal Society Sir Henry Dale fellowship and a Tenure Track fellowship, enabling him to establish his own research group. He moved to the University of Dundee, as Senior Lecturer, in 2023.
Research
Our lab is interested in bacterial infections of the airways. In particular, we seek to understand how the physical, chemical and biological environment of the respiratory tract influences bacterial phenotypes, including those associated with virulence and antimicrobial resistance. Several of the species that we study can act as commensals or asymptomatic colonisers of the upper airways but are also capable of causing severe disease when they descend into the lower regions of the respiratory system. We aim to understand the relationship between commensalism and pathogenesis in these species and to define the host and pathogen factors that drive a switch between the two modes of life.
Much of the work of the lab is centred around Streptococcus pneumoniae, a leading cause of pneumonia, meningitis and sepsis, and Pseudomonas aeruginosa, the major pathogen of the cystic fibrosis lung and a common cause of hospital-acquired infection. Ongoing work examines the process of pathogen adaptation to host environments, using experimental evolution approaches. We also aim to improve on existing in vitro and in vivo models of infection, to better capture the key conditions of the respiratory tract in health and disease. A long-term goal is the development of novel therapeutics and vaccines for treatment and prevention of bacterial respiratory diseases.
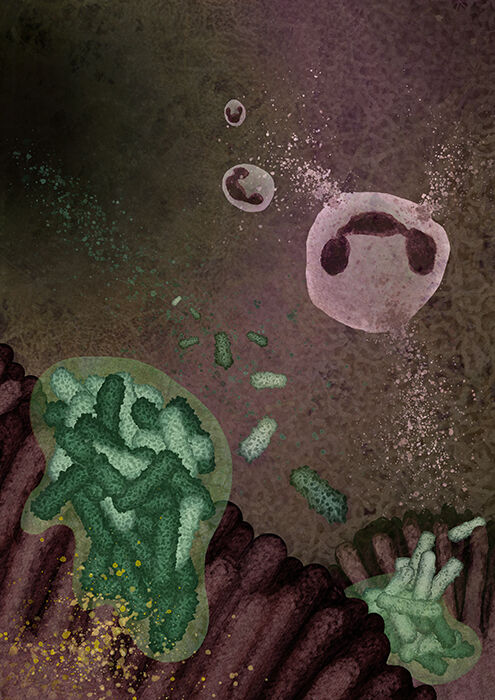
A neutrophil attacking a Pseudomonas aeruginosa biofilm on the airway epithelial surface
Selected Publications
- Secondary messenger signalling influences Pseudomonas aeruginosa adaptation to sinus and lung environments
Ruhluel, D., Fisher, L., Barton, T. E., Leighton, H., Kumar, S., Amores Morillo, P., O'Brien, S., Fothergill, J. L. & Neill, D. R., 22 Apr 2024, The ISME Journal. 18, 1, 15 p., wrae065. - Airway metabolic profiling during Streptococcus pneumoniae infection identifies branched chain amino acids as signatures of upper airway colonisation.
Green, A. E., Pottenger, S., Monshi, M., Barton, T., Phelan, M. & Neill, D. R., 5 Sept 2023, PLoS Pathogens. 19, 9, 30 p., e1011630.
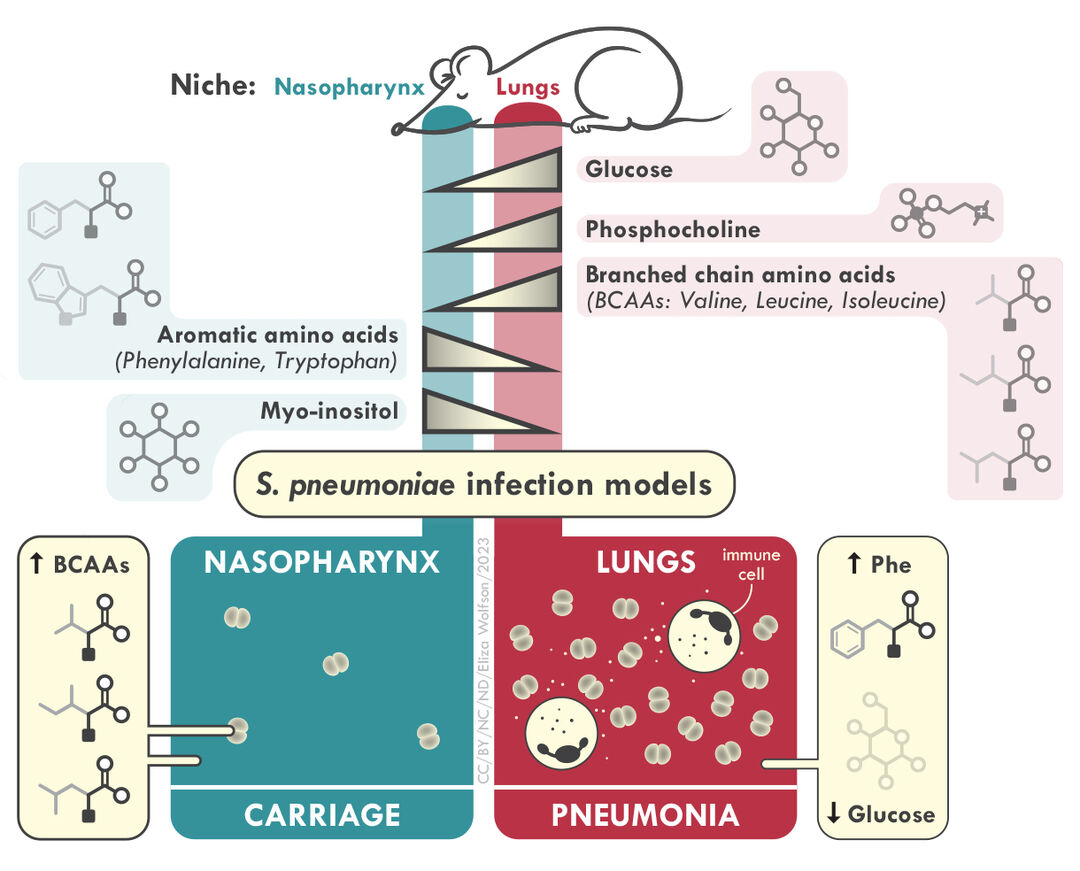
Upper and lower airway microenvironments offer different challenges and opportunities to bacterial pathogens
- An innate pathogen sensing strategy involving ubiquitination of bacterial surface proteins.
Apte S, Bhutda S, Ghosh S, Sharma K, Barton TE, Dibyachintan S, Sahay O, Roy S, Sinha AR, Adicherla H, Rakshit J, Tang S, Datey A, Santra S, Joseph J, Sasidharan S, Hammerschmidt S, Chakravortty D, Oggioni MR, Santra MK, Neill DR, Banerjee A. Sci Adv. 2023 Mar 22;9(12):eade1851. doi: 10.1126/sciadv.ade1851. Epub 2023 Mar 22. PMID: 36947610 - Influence of Streptococcus pneumoniae Within-Strain Population Diversity on Virulence and Pathogenesis.
Jacques LC, Green AE, Barton TE, Baltazar M, Aleksandrowicz J, Xu R, Trochu E, Kadioglu A, Neill DR. Microbiol Spectr. 2023 Feb 14;11(1):e0310322. doi: 10.1128/spectrum.03103-22. Epub 2022 Dec 12. PMID: 36507681 - TNFR2+ regulatory T cells protect against bacteremic pneumococcal pneumonia by suppressing IL-17A-producing γδ T cells in the lung.
Xu R, Jacques LC, Khandaker S, Beentjes D, Leon-Rios M, Wei X, French N, Neill DR, Kadioglu A. Cell Rep. 2023 Jan 30;42(2):112054. doi: 10.1016/j.celrep.2023.112054. PMID: 36724074 - Pseudomonas aeruginosa utilizes the host-derived polyamine spermidine to facilitate antimicrobial tolerance.
Hasan CM, Pottenger S, Green AE, Cox AA, White JS, Jones T, Winstanley C, Kadioglu A, Wright MH, Neill DR, Fothergill JL. JCI Insight. 2022 Nov 22;7(22):e158879. doi: 10.1172/jci.insight.158879. PMID: 36194492
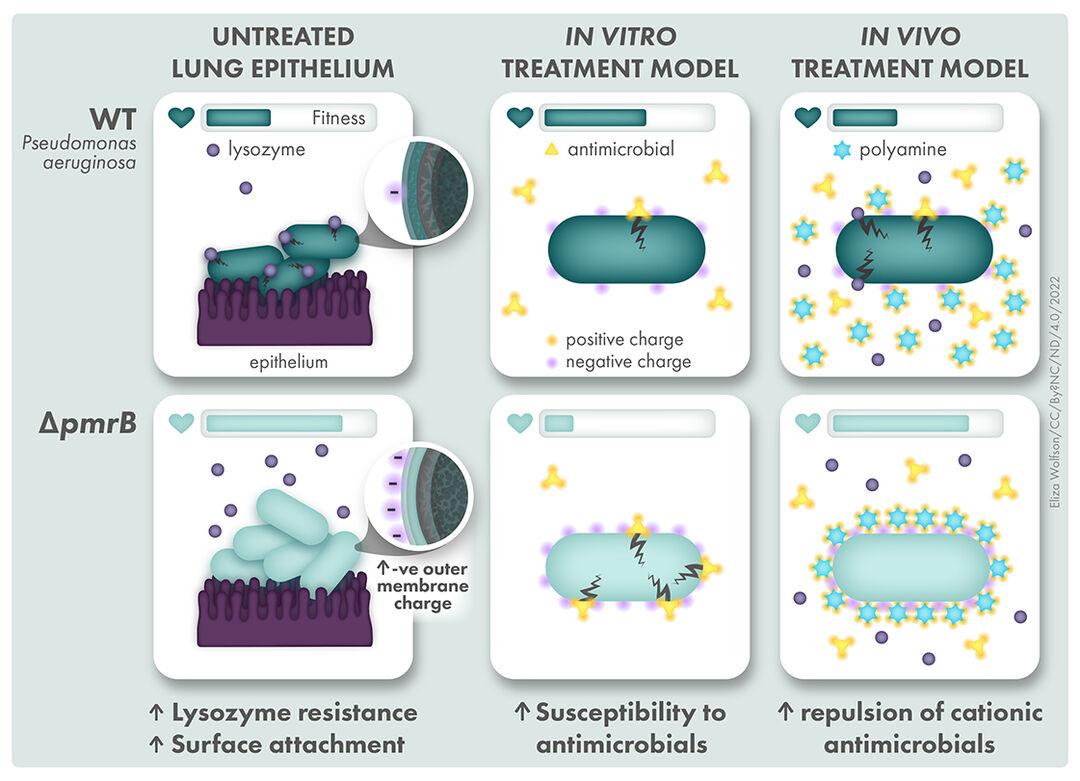
Pseudomonas aeruginosa sequesters host factors to avoid antibiotic killing
- Development of liquid culture media mimicking the conditions of sinuses and lungs in cystic fibrosis and health.
Ruhluel D, O'Brien S, Fothergill JL, Neill DR. F1000Res. 2022 Sep 7;11:1007. doi: 10.12688/f1000research.125074.2. eCollection 2022. PMID: 36519007 - Pneumococcal Colonization and Virulence Factors Identified Via Experimental Evolution in Infection Models.
Green AE, Howarth D, Chaguza C, Echlin H, Langendonk RF, Munro C, Barton TE, Hinton JCD, Bentley SD, Rosch JW, Neill DR. Mol Biol Evol. 2021 May 19;38(6):2209-2226. doi: 10.1093/molbev/msab018. PMID: 33502519 - Structural insights into loss of function of a pore forming toxin and its role in pneumococcal adaptation to an intracellular lifestyle.
Badgujar DC, Anil A, Green AE, Surve MV, Madhavan S, Beckett A, Prior IA, Godsora BK, Patil SB, More PK, Sarkar SG, Mitchell A, Banerjee R, Phale PS, Mitchell TJ, Neill DR, Bhaumik P, Banerjee A. PLoS Pathog. 2020 Nov 20;16(11):e1009016. doi: 10.1371/journal.ppat.1009016. eCollection 2020 Nov. PMID: 33216805 - Influenza-like illness is associated with high pneumococcal carriage density in Malawian children.
Nyazika TK, Law A, Swarthout TD, Sibale L, Ter Braake D, French N, Heyderman RS, Everett D, Kadioglu A, Jambo KC, Neill DR. J Infect. 2020 Oct;81(4):549-556. doi: 10.1016/j.jinf.2020.06.079. Epub 2020 Jul 22. PMID: 32711042
Teaching
- BS42009 – Advanced Molecular Microbiology – module manager and contributor
- BS32004 – Molecular Microbiology - contributor
- Honours Year – project supervisor
- Integrated MSci – project supervisor
PhD Projects
Principal supervisor
Second supervisor
Awards
| Award | Year |
|---|---|
| Wellcome and Royal Society Sir Henry Dale Fellowship | 2017 |
Stories
- Type
- News
Dan Neill joins Molecular Microbiology
Dan Neill has joined the Division of Molecular Microbiology, as Senior Lecturer.