ESCAPE trial information
Information about the ESCAPE trial, what is involved in each stage and how long the trial will take
What is the trial about?
Bronchiectasis is a lung disease that causes people to suffer from cough and chest infections. A bacteria called Pseudomonas causes lung infection in people with bronchiectasis. Pseudomonas is resistant to most antibiotic tablets, which makes it difficult to treat and people can become permanently infected with the bacteria.
People with Pseudomonas infection have more flare ups of their bronchiectasis (exacerbations) where symptoms get worse and more antibiotics are needed. As Pseudomonas infections are difficult to get rid of, preventing them from happening would be better than treating them once they become permanent.
We want to find out whether treating someone with a new infection of Pseudomonas with a course of antibiotics to treat the symptoms, followed by a 3-month course of inhaled antibiotics could get rid of the infection and improve bronchiectasis symptoms.
The trial will compare people with bronchiectasis who receive the trial treatment to people who do not receive the trial treatment.
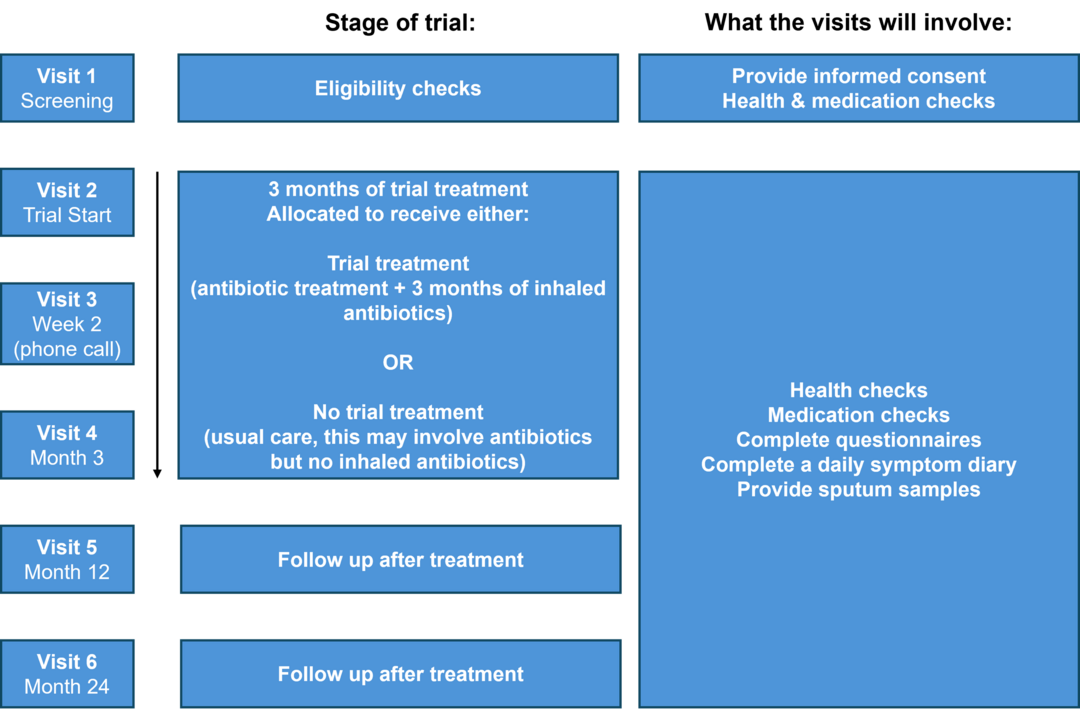
What is involved?
Participation in the trial is entirely voluntary, and you may withdraw at any time. If you join the trial, you will be asked to attend 4 or 5 research visits over 2 years. At each visit you will be seen by a Researcher. During these visits the Researcher will do a simple health check. You will be asked to provide a sputum sample and to complete questionnaires, including a brief daily symptom diary (throughout the trial). Taxi transport can be arranged, or travel expenses reimbursed.
You will be allocated to receive either the trial treatment or no trial treatment. You will not be able to choose which treatment you receive; this will be decided randomly. You and the research team will know which treatment you have been allocated.
Trial treatment: you’ll be given antibiotics (either oral or drip), followed by 3-months of an inhaled antibiotic treatment through a nebuliser machine.
No trial treatment: you’ll receive the usual care from your doctor. This may involve a short course of antibiotics if you have symptoms of infection.
What are the possible benefits of taking part?
By taking part you are contributing to medical science. The main benefit of taking part will be to see whether long-term inhaled antibiotics could benefit you and future patients with bronchiectasis.
What are the possible disadvantages of taking part?
If you receive the trial treatment, your doctor will prescribe you antibiotics which are commonly used in people with bronchiectasis and are recommended in the British Thoracic Society guidelines. All medications have the potential to have some side effects; you will receive a patient leaflet with information on how to take the antibiotics and any known side effects.
Will my taking part in the trial be kept confidential?
In this research trial we will use information from you and your medical records. We will only use information that we need for the research trial. We will let very few people know your name or contact details, and only if they really need it for this trial. Everyone involved in this trial will keep your data safe and secure. We will also follow all privacy rules.
At the end of the trial, we will save some of the data in case we need to check it and for future research. We will make sure no-one can work out who you are from the reports we write.
The Participant Information Sheet tells you more about this.